The FDA’s approval process has long been criticized. Generic drugs and devices frequently reach the market simply because their makers can show that they are comparable to previously approved goods, even if those items have well-known safety issues. Although some medicines may present a danger, the FDA has found that their advantages outweigh the potential risks.
Contrary to popular belief, just because a medicine or technology is FDA-approved does not imply it is guaranteed safe. Manufacturers withdraw around 4,500 drugs and medical devices every year. While the FDA has procedures in place to prevent these safety concerns, there are still times when new medicines or equipment may appear rapidly, resulting in safety issues.
The FDA Medical Device Approval Procedure
The FDA is responsible for reviewing medical devices as well. The study of medical equipment begins in the laboratory. Most gadgets are subjected to basic testing in the lab and on animals to establish their safety and efficacy, after which they are tested on people to verify that they are safe and effective.
Most gadgets go through animal testing first to answer fundamental safety concerns. The FDA then reviews all the submitted information and determines whether or not to approve it. The FDA divides medical devices into three categories based on their risk potential.
The lowest-risk devices include oxygen masks and surgical instruments. Class III gadgets help or sustain life, are inserted into the body, or have the potential for significant illness or injury. These include cardiac pacemakers, breast implants, and HIV diagnostic tests.
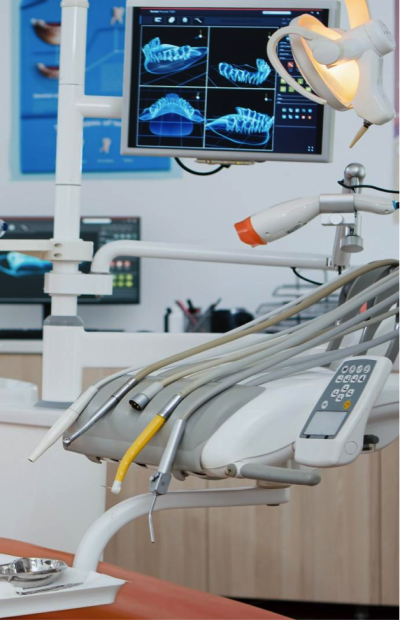 Medical devices are frequently able to move between classes based on the results of medical research.
Medical devices are frequently able to move between classes based on the results of medical research.
Types of Medical Device Applications
There are numerous types of applications for medical devices, depending on the level of risk a device presents. The approval procedure for prescription drugs is similar to that for medical devices.
The FDA (or an equivalent entity) sends a premarket notification, also known as PMN or 510(k), to devices categorized as low- to moderate risk. According to federal law, new device manufacturers must register with the FDA and notify the agency at least 90 days before selling their goods. The premarket notification must show that the device is as safe and effective as a legally marketed device and practically identical. There is no requirement for clinical research.
Premarket approval is required for high-risk products, which are the most rigorous kind of device application administered by the FDA. This application is for Class III devices that were found to be less than substantially equivalent to a Class I or II marketed device. In this instance, sufficient scientific proof must be presented to show that the device is safe and effective for its stated purpose.
A Humanitarian Use Device is a device that is intended to benefit patients by treating or diagnosing a disease or condition that affects fewer than 8,000 people in the United States each year. This application is comparable to a premarket approval application, but pharmaceutical firms are not required to show efficacy.
Medical Device Critiques
Critics have lambasted the FDA for failing to keep up with changing technology. That’s because numerous medical devices have been recalled after it was found that they were potentially harmful.
Critics have also expressed concerns about the FDA’s monitoring of medical devices after release. They worry that inadequate oversight might allow issues to go unnoticed.
Drug Patents
 Before brand-name manufacturers’ patents expire, generic drug producers are permitted by federal law to work on drugs to receive FDA approval. When a generic-drug maker files an FDA application, it must notify the patent holder if it is contesting the brand’s patent, implying that the generic drug does not infringe on the brand patent or that the invention isn’t valid.
Before brand-name manufacturers’ patents expire, generic drug producers are permitted by federal law to work on drugs to receive FDA approval. When a generic-drug maker files an FDA application, it must notify the patent holder if it is contesting the brand’s patent, implying that the generic drug does not infringe on the brand patent or that the invention isn’t valid.
Suppose the brand-name firm fails to sue the generic-drug producer within 45 days. In that case, the FDA cannot approve the generic drug for 30 months unless the patent expires or is judged invalid or not infringed before or until that generic-drug company wins in court.
In essence, it has formed a legal no man’s land in which companies are engaged in a patent battle. If a generic-drug producer markets a product and breaches the patent, it might be required to pay the brand-name firm compensation.
Reasons that Approval May Be Delayed or Refused
The FDA provides a response letter to the pharmaceutical firm that submitted the application when it rejects a drug approval. The agency allows the drug maker to meet with FDA officials about any concerns. The company has three options: ask for a hearing, make necessary adjustments, submit new data, or withdraw the application.
The FDA has a rigorous process for approving medical devices. This process includes several types of applications, reviews by experts, and the possibility of delay or refusal. Mr. Letko Edward is an expert in this process and can help your company get its device approved quickly and efficiently. Visit our website for information on this and much more!

